

 FROM
UNIT CELL TO LATTICE
FROM
UNIT CELL TO LATTICE
If we take the square unit cell S2 and stack it, we produce this
square lattice. Notice that once we begin stacking the unit cells, we never
change the orientation of any subsequent unit cells as they stack.
In other words, once the orientation of a unit cell is determined, all
unit cells within that lattice have the same orientation.


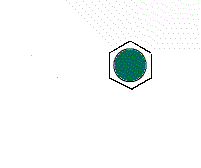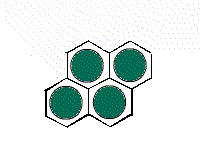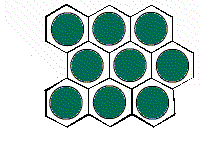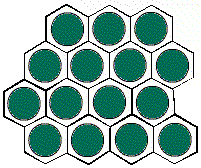
 The hexagonal unit
cell H1 gives a different arrangement of circles.
The hexagonal unit
cell H1 gives a different arrangement of circles.
 `
`
In the figures below we have created lattices from unit cells. It
is the stacking which creates lines and cavities which are not immediately
obvious from looking at the unit cells.
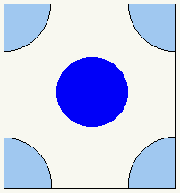



 S1 unit cell
S1 lattice
S1 unit cell
S1 lattice
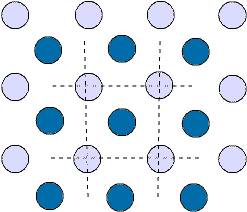
We can look at the S1 lattice as alternating staggered rows of solid
and empty circles - both horizontally and vertically.
We can also see it as diagonal rows of alternating circles.
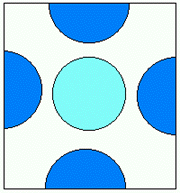

 S3 unit cell
S3 lattice
S3 unit cell
S3 lattice
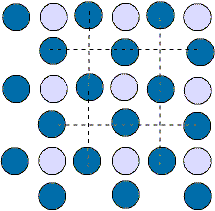
We can see lattice S3 as different diagonal lines of atoms, or horizontal
rows, or vertical columns. If we rotate the entire lattice 90 degrees we
see an identical pattern. This is a property of a square lattice.




 H1
lattice
H1
lattice
 In the H1 lattice we can view the structure as parallel lines of circles
along the X - axis, each line staggered from the next.
In the H1 lattice we can view the structure as parallel lines of circles
along the X - axis, each line staggered from the next.
If we rotate the entire lattice 60 degrees or 120 degrees
we see an identical pattern. This is because the unit cell is hexagonal.

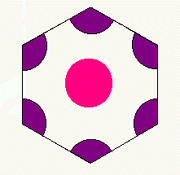

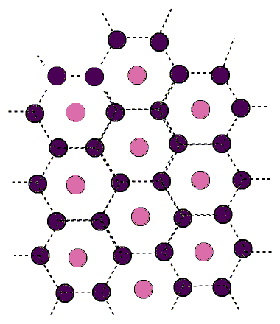
 H3
lattice
H3
lattice
In the H3 lattice, the pattern of lines is the same (every 60 degrees),
but each line contains the repeating sequence (pink- purple-purple).
Click
here to go to the next page.
Structure of Crystals
Crystal Lattices
Unit Cells
 From
Unit Cell to Lattice
From
Unit Cell to Lattice From Lattice to Unit Cell
Stoichiometry
Packing & Geometry
Simple Cubic Metals
Close Packed Structures
Body Centered Cubic
Cesium Chloride
Sodium Chloride
Rhenium Oxide
Niobium Oxide
Except as otherwise noted, all images and movies
are owned and copyright
From Lattice to Unit Cell
Stoichiometry
Packing & Geometry
Simple Cubic Metals
Close Packed Structures
Body Centered Cubic
Cesium Chloride
Sodium Chloride
Rhenium Oxide
Niobium Oxide
Except as otherwise noted, all images and movies
are owned and copyright  by
Dr. Barbara L. Sauls and Dr. Frederick C. Sauls
1998.
Contact the owners for individual permission
to use. barbarasauls@kings.edu
by
Dr. Barbara L. Sauls and Dr. Frederick C. Sauls
1998.
Contact the owners for individual permission
to use. barbarasauls@kings.edu









