 THE
STRUCTURE OF CRYSTALS
THE
STRUCTURE OF CRYSTALS
The beauty and symmetry of crystals have fascinated people for thousands
of years. Somehow, they seem different from more ordinary matter. However,
most solid objects are made of many tiny interlocking crystals. It is only
occasionally that the individual crystals are large enough to catch our
attention, as in these beautiful examples.




 Celestite
Sulfur
Pyrite
The above images are the property of Amethyst
Galleries, Inc., and may not be copied for commercial purposes.
Permission to copy descriptions and images is
granted for personal and educational use only.
Refer to http://mineral.galleries.com/
Celestite
Sulfur
Pyrite
The above images are the property of Amethyst
Galleries, Inc., and may not be copied for commercial purposes.
Permission to copy descriptions and images is
granted for personal and educational use only.
Refer to http://mineral.galleries.com/
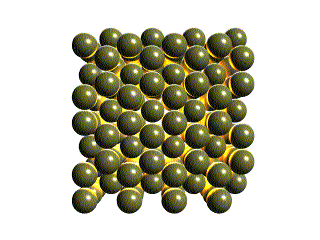
 The atoms in a crystal are in a regular repeating pattern called the
crystalline lattice. Although the existence of this lattice
has been known for many years, recently the scanning tunneling microscope
(STM) was invented. This allows us to see individual atoms. Below is an
STM image of a platinum surface. Note the regular rows of Pt atoms.
The atoms in a crystal are in a regular repeating pattern called the
crystalline lattice. Although the existence of this lattice
has been known for many years, recently the scanning tunneling microscope
(STM) was invented. This allows us to see individual atoms. Below is an
STM image of a platinum surface. Note the regular rows of Pt atoms.


 This image is the property of IBM Corporation.
This image is the property of IBM Corporation.

The lattice can have a profound influence on the material's properties.
For example, below we see two crystals - graphite and diamond. They each
contain only 1 kind of atom - carbon. The difference is in how those atoms
are arranged.


 Graphite
Diamond
The above images are the property of Amethyst
Galleries, Inc., and may not be copied for commercial purposes.
Permission to copy descriptions and images is
granted for personal and educational use only.
Refer to http://mineral.galleries.com/
Graphite is black, soft, and an excellent lubricant. This suggests that
it must be easy to separate atoms in graphite, or at least to make them
slide past one another.
Graphite
Diamond
The above images are the property of Amethyst
Galleries, Inc., and may not be copied for commercial purposes.
Permission to copy descriptions and images is
granted for personal and educational use only.
Refer to http://mineral.galleries.com/
Graphite is black, soft, and an excellent lubricant. This suggests that
it must be easy to separate atoms in graphite, or at least to make them
slide past one another.
Diamond is transparent, strong, and very hard. It is a superb cutting
tool. The atoms in diamond must be very strongly held in position.
Chemically, diamond is inert. Graphite, on the other hand, is very
useful for absorbing and catalyzing reactions. The structures of
diamond and graphite explain these differences.
The carbon atoms in diamond are each bound to 4 others in a 3-dimensional
network. To change the shape of a diamond or break it requires that many
of these strong bonds be broken simutaneously. Since there is little room
for other molecules to get into the structure, diamond is not very reactive.
Click on the image below to view the structure of diamond.
 Graphite consists of sheets of strongly bonded hexagonal rings. Each
sheet is far from and weakly bound to the next. Thus, the sheets can slide
past one another, making graphite a soft lubricant. The spaces between
the layers allow other molecules to enter, explaining the absorbing and
catalytic properties.
Click on the image below to view the structure of graphite.
Graphite consists of sheets of strongly bonded hexagonal rings. Each
sheet is far from and weakly bound to the next. Thus, the sheets can slide
past one another, making graphite a soft lubricant. The spaces between
the layers allow other molecules to enter, explaining the absorbing and
catalytic properties.
Click on the image below to view the structure of graphite.
 The structures of graphite and diamond have long been known. However
only recently has the scanning tunneling microscope (STM) given visual
confirmation.
The structures of graphite and diamond have long been known. However
only recently has the scanning tunneling microscope (STM) given visual
confirmation.
Shown below is an STM image of a graphite surface. This is an actual
image, not a simulation. It clearly shows the interconnected 6-membered
rings of graphite and the triangular geometry about each carbon atom.
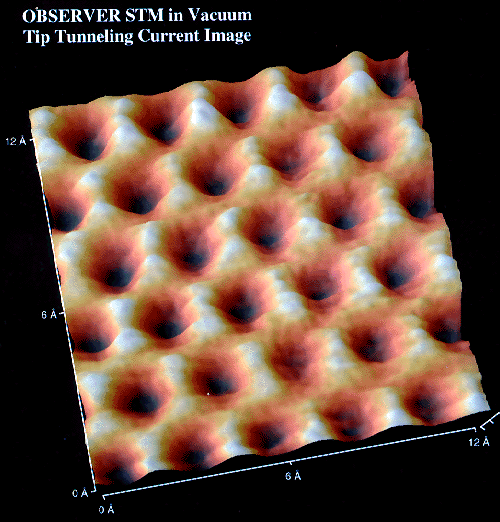 Used with permission TopoMetrix Corporation
Used with permission TopoMetrix Corporation  5403 Betsy Ross Drive, Santa Clara, CA
95054
Boron nitride also exists in two different crystalline structures.
Click on each image below to view that structure.
5403 Betsy Ross Drive, Santa Clara, CA
95054
Boron nitride also exists in two different crystalline structures.
Click on each image below to view that structure.
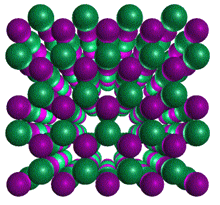
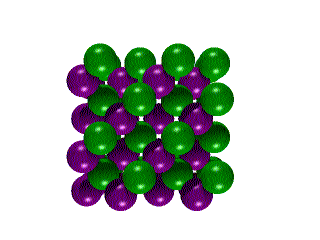 Hexagonal Boron Nitride
Hexagonal Boron Nitride Cubic
Boron Nitride
Cubic
Boron Nitride Click
here to go to next page.
Click
here to go to next page.
 Structure
of Crystals
Structure
of Crystals Crystal Lattices
Unit Cells
From Unit Cell to Lattice
From Lattice to Unit Cell
Stoichiometry
Packing & Geometry
Simple Cubic Metals
Close Packed Structures
Body Centered Cubic
Cesium Chloride
Sodium Chloride
Rhenium Oxide
Niobium Oxide
Crystal Lattices
Unit Cells
From Unit Cell to Lattice
From Lattice to Unit Cell
Stoichiometry
Packing & Geometry
Simple Cubic Metals
Close Packed Structures
Body Centered Cubic
Cesium Chloride
Sodium Chloride
Rhenium Oxide
Niobium Oxide
Except as otherwise noted, all images and movies
are owned and copyright  by
Dr. Barbara L. Sauls and Dr. Frederick C. Sauls
1998.
Contact the owners for individual permission
to use. barbarasauls@kings.edu
by
Dr. Barbara L. Sauls and Dr. Frederick C. Sauls
1998.
Contact the owners for individual permission
to use. barbarasauls@kings.edu










