We may consider a crystal as a collection of identical unit cells. For a substance containing more than 1 kind of atom, the ratio of atoms in the unit cell must be exactly the same as in the entire crystal.
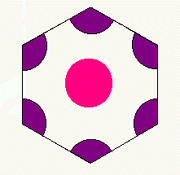
For example, the unit cell H3 contains 1 pink and (6 x 1/3 = 2) purple
circles, or a ratio of 1:2.
![]()
H3 unit cell
H3 lattice
The full lattice must also have a 1:2 ratio. Because the drawing
does not end at the edges of the unit cells, the ratio you see (10:30)
is not representative.