
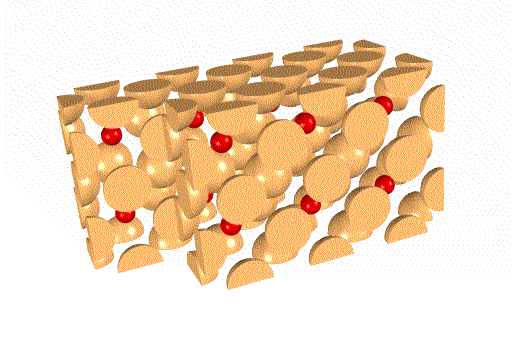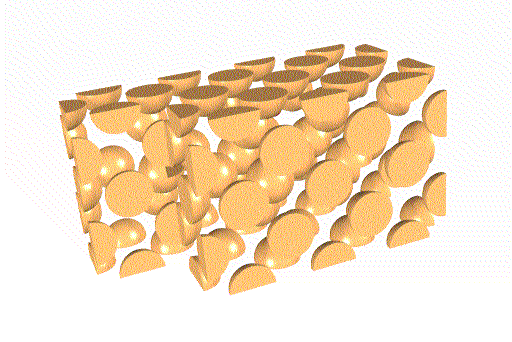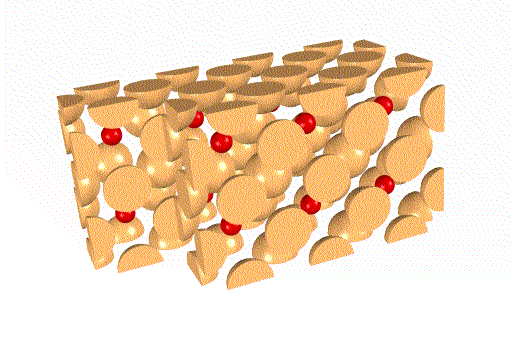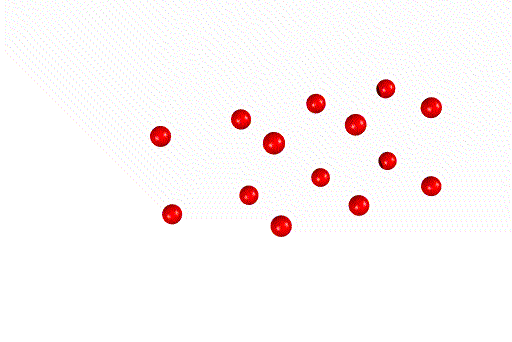
RHENIUM OXIDE (ReO3)
Rhenium oxide forms a more complex and interesting
structure.
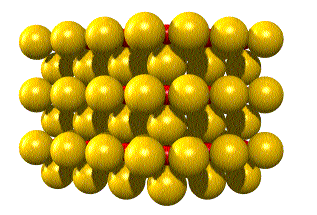
In the unit cell below, rhenium (Re) is red
and oxygen (O) is yellow.

Click on the unit cell above to explore it in VRML.
The unit cells stack to form the crystal thus:

 Click
on the Rhenium Oxide lattice to
Click
on the Rhenium Oxide lattice to Click
on the expanded ReO3 lattice
Click
on the expanded ReO3 lattice
view the expanded version in rotation. structure
to explore it in VRML.
structure
to explore it in VRML.


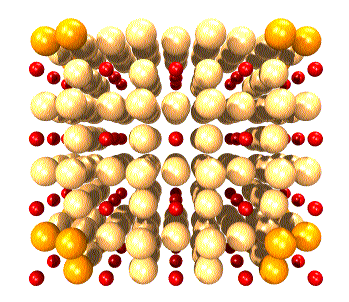

The oxygen ions at the corners of the expanded lattice have been
made darker simply to help keep the structure in perspective. They are
otherwise identical to all other oxygens in the lattice.
Particularly interesting are:
(1) In some orientations square boxes of oxygen atoms appear, filled
with columns of rhenium. These are easier to see in the open
lattice.
(2) In other orientations similar, but empty square channels exist.
These can allow other atoms, molecules, or ions to migrate within the crystal.
This provides a mechanism for the crystal to act as a catalyst or for the
crystal to change its composition without breaking down the lattice itself.
The ReO3 sublattices are not identical, as you can see
below.
Click
here to go to the next page.

Structure of Crystals
Crystal Lattices
Unit Cells
From Unit Cell to Lattice
From Lattice to Unit Cell
Stoichiometry
Packing ? Geometry
Simple Cubic Metals
Close Packed Structures
Body Centered Cubic
Cesium Chloride
Sodium Chloride
 Rhenium
Oxide
Rhenium
Oxide
Niobium Oxide

 Except
as otherwise noted, all images, movies and VRMLs are owned and copyright
Except
as otherwise noted, all images, movies and VRMLs are owned and copyright  by
by
Barbara L. Sauls and Frederick C. Sauls 2000.
Contact the owners for individual permission
to use. blsauls@kings.edu


![]() Click
on the Rhenium Oxide lattice to
Click
on the Rhenium Oxide lattice to![]() Click
on the expanded ReO3 lattice
Click
on the expanded ReO3 lattice
![]() structure
to explore it in VRML.
structure
to explore it in VRML.

![]()
![]()

![]()
